 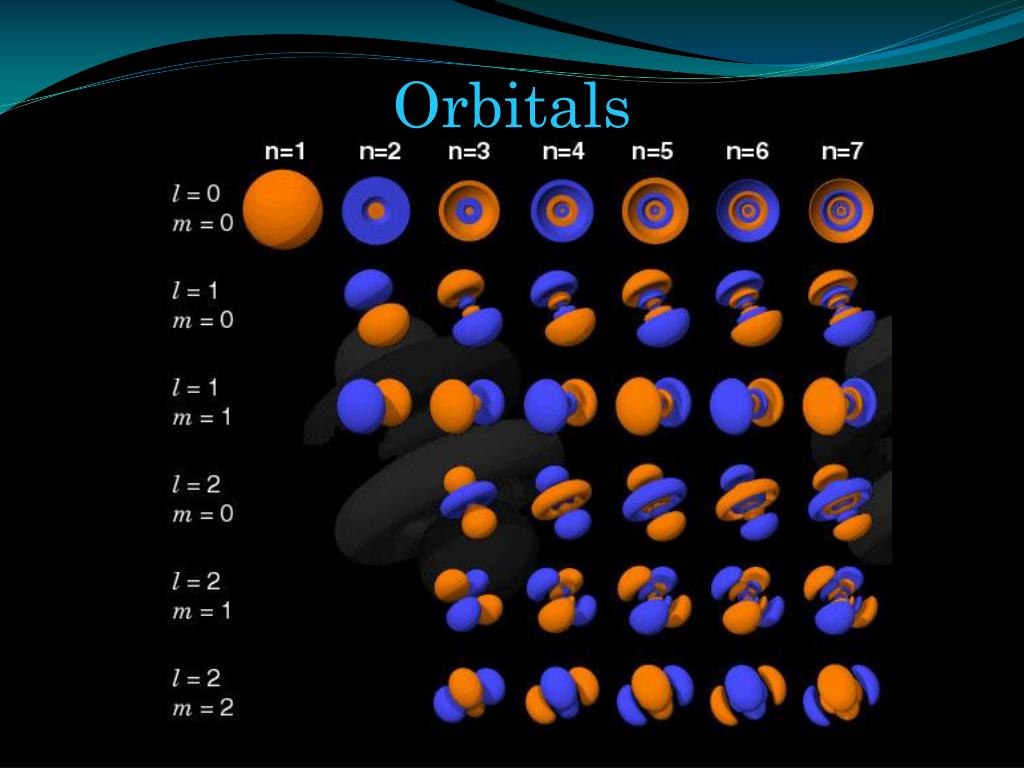
There are only two possible values of the spin quantum number: -1/2 and +1/2. Spin Quantum Number: indicates the direction in which an electron in an orbital is spinning.Because different orbitals have different shapes, they can be rotated around the a 3D-coordinate plane many orientations. Magnetic Quantum Number ( m): indicates the orientation of an orbital around the nucleus.This will be discussed more in detail later. Different values of l correspond to specific shapes of electron orbitals. Angular Momentum Quantum Number ( l): indicates the shape of an orbital.The higher the principal quantum number is, the higher the energy level, and therefore the more the distance is between the nucleus and the particular electron. Principal Quantum Number ( n): indicates main energy level of an electron.Basic Review of the Quantum Numbersįor more details on the quantum numbers, check out our quantum numbers article! There are four quantum numbers that tell the energy level, shape, orientation, and electron spin of orbitals. 
For example, the 5 orientations of d-orbitals on the 3D-plane are degenerate orbitals. “Atomic orbital” generally refers to the region of space where an electron could possibly be present, based on the mathematical function for that orbital.ĭegenerate orbitals are orbitals in the same main energy level and sublevel that have different orientations. The precise definition of an orbital, is a mathematical function that describes the location of an electron in an atom, based on probability. We will learn about the s orbital, p orbital, d orbital and f orbital. Atomic orbitals come in different shapes, depending on the number of electrons the atom has. Electron orbitals are the three-dimensional areas around the nucleus of an atom where a particular electron resides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
